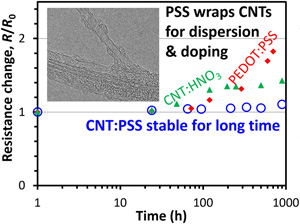
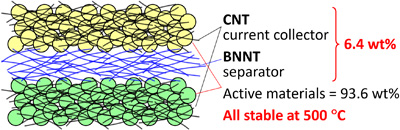
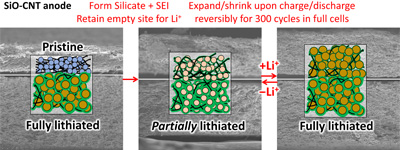
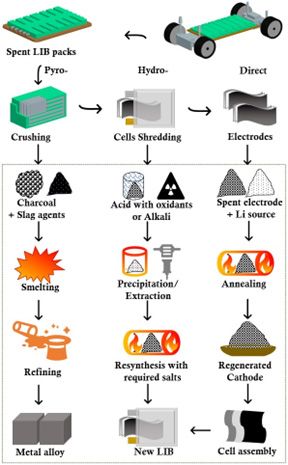
On the other hand, as their price (higher than gold) shows, their fabrication process is still under development and their practical applications are very much limited. Chemistry & engineering should lead the innovations for their production and manufacturing. We have developed rapid growth process of millimeter-long single-wall CNTs and are trying to realize their practical production. Please click here for details.
We are developing mass-production processes of CNTs by utilizing three-dimensional space of reactors, and direct fabrication of various devices by growing CNTs on device substrates. We have also started the synthesis of boron nitride nanotubes (BNNTs) having similar structure as CNTs and insulative property.
- Pengfei CHEN (D3): Activation of alkane for CVD synthesis of CNTs.
- Koyo ANDO (M2): Synthesis of CNTs by floating catalyst CVD method and development of transparent thin films.
- Koki AKIYAMA (M1): Fluidized-bed synthesis of long and pure CNTs, their diameter/wall-number control and applications to batteries.
- Yoshiyuki MATSUKAWA (M1): Co-production of CNTs and low-carbon fuels.
- Haruto TOMODA (B4): BN coating of single-wall CNTs for battery applications.
- Nonoka NEGISHI (B4): Long-term continuous gas-phase synthesis of high-quality single-wall CNTs.
- Kazuma YAMAMOTO (B4): Purification and BN coating of CNTs for battery applications.

Rapid SWCNT growth [58]. Larger Movie.

Continuous production by fluidized bed [60].
Watch detailed version here.